Basic Chemistry
I. THE ATOM
A. Atom - smallest unit of matter unique to a particular element.
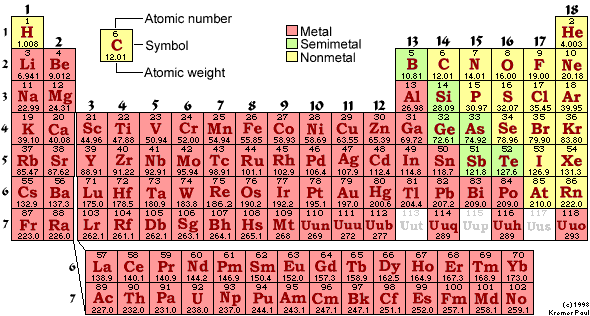
B. Element - A substance made up of only one kind of atom - ex. carbon contains only carbon atoms. Elements can't be broken down into other substances by ordinary chemical means. Each element displays unique properties (ex. some are gases, some are solids, some are metals, etc.). About 92 elements occur naturally (there are also some man-made elements). Some of the elements important to our study of living systems are carbon, oxygen, hydrogen, nitrogen, sodium, chlorine, phosphorus, and potassium. You may also be familiar with the elements lead, iron, iodine, gold, silver, and copper, nickel, and platinum.
C. Some Important Things to Know about Atoms & Elements:
1. An atom consists of 2 basic parts:
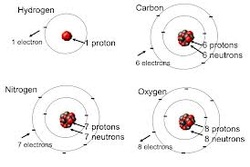
a. nucleus - the nucleus contains protons & neutrons:
1.) protons - positively charged; all atoms have protons; protons give the nucleus a positive charge.
2.) neutrons - neutral in charge; fxn.: "stabilizers"; all atoms except hydrogen have one or more neutrons.
b. electrons – negatively charged; occupy orbit energy levels or shells around the nucleus; attracted to the positive charge of the nucleus; in an atom, the number of electrons always equals the number of protons, so the atom, as a whole, has no charge; electrons determine the chemical properties of elements (ex. whether they are a liquid, solid, or gas, etc.).
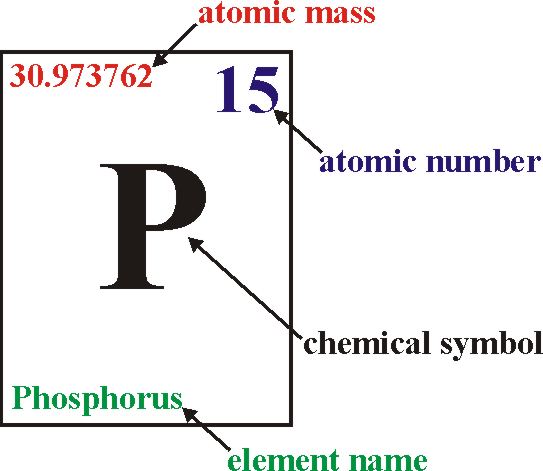
2. atomic number = number of protons (or number of electrons); in an atom, the number of protons always equals the number of electrons; this number differs for each element.
3. atomic mass number = number of protons + number of neutrons; the number of electrons is not included in the mass number due to their insignificant mass.
4. The 6 elements important for building organic molecules like carbohydrates, lipids, proteins, and nucleotides are: carbon, hydrogen, oxygen, sulfur, nitrogen, & phosphorus.
Note: Be able to determine the number of electrons, protons, & neutrons in an atom, given the atomic number & atomic mass. Ex. Sodium (Na) has an atomic number of 11 & an atomic mass of 23. A Sodium atom has 11 protons, 11 electrons, & 12 neutrons.
source for Chemistry summary of Cowan Chapter 2: http://www.austincc.edu/rohde/CHP2.HTM
A. Atom - smallest unit of matter unique to a particular element.
B. Element - A substance made up of only one kind of atom - ex. carbon contains only carbon atoms. Elements can't be broken down into other substances by ordinary chemical means. Each element displays unique properties (ex. some are gases, some are solids, some are metals, etc.). About 92 elements occur naturally (there are also some man-made elements). Some of the elements important to our study of living systems are carbon, oxygen, hydrogen, nitrogen, sodium, chlorine, phosphorus, and potassium. You may also be familiar with the elements lead, iron, iodine, gold, silver, and copper, nickel, and platinum.
C. Some Important Things to Know about Atoms & Elements:
1. An atom consists of 2 basic parts:
a. nucleus - the nucleus contains protons & neutrons:
1.) protons - positively charged; all atoms have protons; protons give the nucleus a positive charge.
2.) neutrons - neutral in charge; fxn.: "stabilizers"; all atoms except hydrogen have one or more neutrons.
b. electrons – negatively charged; occupy orbit energy levels or shells around the nucleus; attracted to the positive charge of the nucleus; in an atom, the number of electrons always equals the number of protons, so the atom, as a whole, has no charge; electrons determine the chemical properties of elements (ex. whether they are a liquid, solid, or gas, etc.).
2. atomic number = number of protons (or number of electrons); in an atom, the number of protons always equals the number of electrons; this number differs for each element.
3. atomic mass number = number of protons + number of neutrons; the number of electrons is not included in the mass number due to their insignificant mass.
4. The 6 elements important for building organic molecules like carbohydrates, lipids, proteins, and nucleotides are: carbon, hydrogen, oxygen, sulfur, nitrogen, & phosphorus.
Note: Be able to determine the number of electrons, protons, & neutrons in an atom, given the atomic number & atomic mass. Ex. Sodium (Na) has an atomic number of 11 & an atomic mass of 23. A Sodium atom has 11 protons, 11 electrons, & 12 neutrons.
source for Chemistry summary of Cowan Chapter 2: http://www.austincc.edu/rohde/CHP2.HTM